 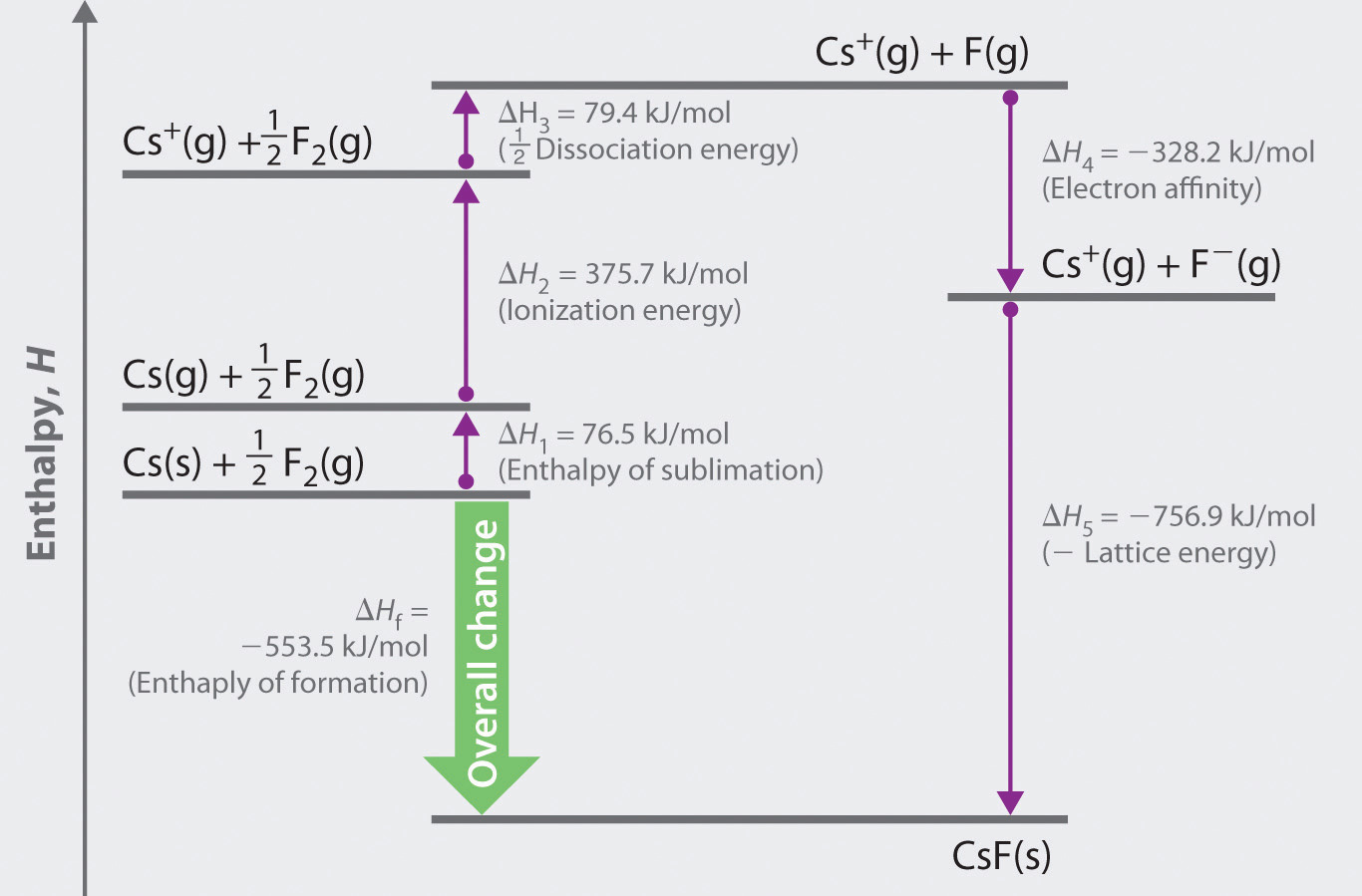
One of the key Lattice Enthalpy questions is the two principal factors that affect it. What are the Factors of Lattice Enthalpy?Īs you know by now, Lattice Enthalpy is the change in enthalpy associated with the formation of one mole of an ionic compound from its gaseous ions under normal conditions. So, the breaking up and the formation both get a reference as Lattice Enthalpy. In the case of sodium chloride case, it would be +787kJ mol -1. The second definition describes it as the enthalpy when 1 mole of sodium chloride (or any other substance) is broken up to form its scattered gaseous ions. In the case of sodium chloride case, that would be -787kJ mol -1. We can say that the enthalpy is the change when 1 mole of sodium chloride (or any other substance) was formed from its scattered gaseous ions. We can describe Lattice Enthalpy in the following two ways: Lattice is a strong network, mesh, or web and enthalpy is the heat content, total heat, or a thermodynamic quantity equal to the internal energy of a system and the product of its volume and pressure. We can even say that Lattice Enthalpy is a measure of the strength of an ionic compound. 
It is the energy required to entirely separate one mole of an ionic compound into gaseous ions. In simple words, Lattice Enthalpy is the change in Enthalpy connected with the formation of one mole of an ionic compound from its gaseous ions, other things remaining standard. Lattice Enthalpy is the change when this structure is formed or is broken. 
The strong bonds between oppositely charged ions lock them into a network or lattice within the crystalline structure. Ionic compounds are solids that have high boiling and melting points. It takes enough energy to break their bond. The crystalline structure of ionic compounds is strong and rigid. In this article, we shall discuss ionic solids and what Lattice Enthalpy is. A chemical reaction is a process that involves a rearrangement of the molecular or ionic structure of a substance. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
